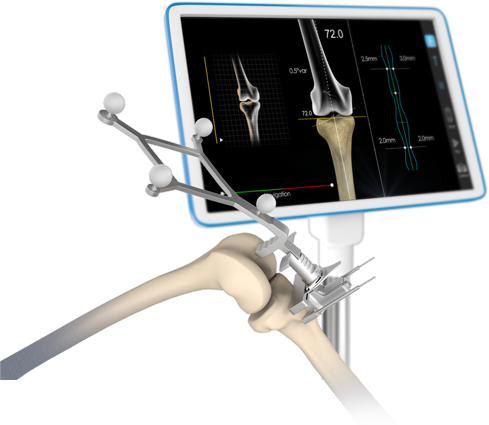
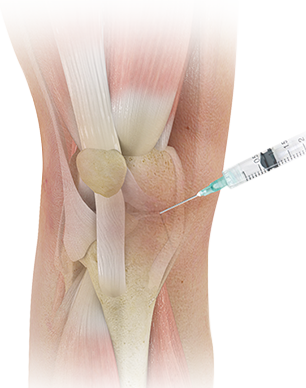
Specialties
Dr. Meftah is a highly trained and experienced orthopedic surgeon specializing in hip and knee reconstruction, as well as diagnosing and treating arthritis. Click on the below tabs to learn more about his services.
- Knee
Replacement - Revision Knee
Replacement - Hip
Replacement - Revision Hip
Replacement - Total Hip
Replacement - Hip
Hemiarthroplasty - Total Knee
Replacement - Partial Knee
Replacement - Minimally Invasive
Hip Replacement - Outpatient
Hip Replacement - Outpatient
Knee Replacement - Rapid Recovery
Knee Replacement - Rapid Recovery
Hip Replacement - Periprosthetic
Hip Fractures - Periprosthetic
Knee Fractures
Meet Morteza Meftah, M.D. Board Certified Hip and Knee Joint Reconstruction Specialist Co-director of Robotic and Computer-assisted Joint Replacement SurgeryChair, Center for Orthopedic Innovation, Department of Orthopedic Surgery
My passion for orthopedic surgery started during medical school, when I experienced how much a hip and knee replacement can improve a patient’s quality of life.
At NYU Langone’s Preston Robert Tisch Center for Men’s Health and NYU Langone Madison Avenue Orthopedics, I specialize in hip and knee reconstruction, as well as diagnosing and treating arthritis...
AREAS OF EXPERTISE
Publications
All-Polyethylene Tibial Implant in Young, Active Patients: A Concise Follow-Up, 10 to 18 years
Abstract This is a long-term follow-up report of all-polyethylene tibial components in 60 years and younger, active patients. At the mean follow-up of 12.4 ± 2.7 (range, 10-18 years), no more patients...